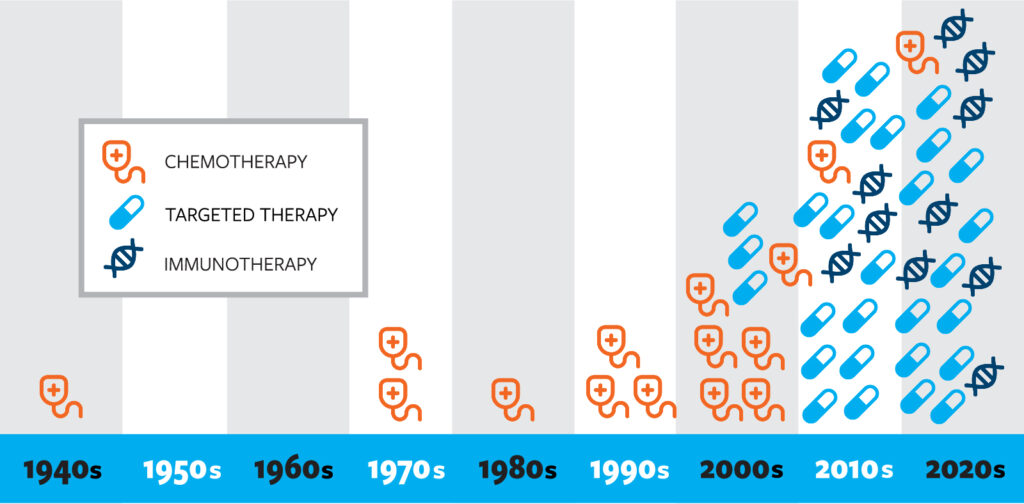
The following chart demonstrates how the pace of FDA approvals for lung cancer treatments has accelerated in recent decades. This rapid development is a result of scientific discoveries and continued progress made possible by funding research. See a list of drugs by decade
FDA approvals in the 2020s
Below is a list of recent FDA approvals for the treatment of lung cancer. In a relatively short time, we’re seeing an exponential increase in treatment approvals!
2023
October
- The FDA has approved encorafenib (Braftovi®) plus binimetinib (Mektovi®) for adult patients with metastatic non–small cell lung cancer harboring a BRAF V600E mutation. Read more
August
- FDA granted regular approval to pralsetinib (Gavreto®) for adult patients with metastatic rearranged during transfection (RET) fusion-positive non-small cell lung cancer (NSCLC).
January
- FDA approved pembrolizumab (Keytruda®) for adjuvant treatment following resection and platinum-based chemotherapy for stage 1B (T2a ≥4 cm), 2, or 3A non-small cell lung cancer (NSCLC).
2022
December
- FDA granted accelerated approval to adagrasib (Krazati®), a RAS GTPase family inhibitor, for adult patients with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) who have received at least one prior systemic therapy.
November
- FDA approved cemiplimab-rwlc (Libtayo®) in combination with platinum-based chemotherapy for adult patients with advanced non-small cell lung cancer (NSCLC) with no EGFR, ALK, or ROS1 aberrations.
- FDA approved tremelimumab (Imjudo®) in combination with durvalumab (Imfinzi®) and platinum-based chemotherapy for adult patients with metastatic non-small cell lung cancer (NSCLC) with no sensitizing epidermal growth factor receptor (EGFR) mutation or anaplastic lymphoma kinase (ALK) genomic tumor aberrations.
September
- FDA granted regular approval to selpercatinib (Retevmo®) for adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with a rearranged during transfection (RET) gene fusion.
August
- FDA granted regular approval to capmatinib (Tabrecta®) for adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors have a mutation leading to mesenchymal-epithelial transition (MET) exon 14 skipping.
- FDA granted accelerated approval to fam-trastuzumab deruxtecan-nxki (Enhertu®) for adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating human epidermal growth factor receptor 2 HER2 (ERBB2) mutations, and who have received a prior systemic therapy. This is the first drug approved for HER2-mutant NSCLC.
March
- FDA approved nivolumab (Opdivo®) with platinum-doublet chemotherapy for adult patients with resectable non-small cell lung cancer (NSCLC) in the neoadjuvant setting.
2021
October
- FDA approved atezolizumab (Tecentriq®) for adjuvant treatment following resection and platinum-based chemotherapy in patients with stage 2 to 3A non-small cell lung cancer (NSCLC) whose tumors have PD-L1 expression on ≥ 1% of tumor cells.
September
- FDA approved mobocertinib (Exkivity®) for the treatment of adult patients with locally advanced or metastatic non–small cell lung cancer with EGFR exon 20 insertion mutations whose disease has progressed on or after platinum-based chemotherapy. Read more
May
- FDA approved amivantamab-vmjw (Rybrevant™) for adults with NSCLC with epidermal growth factor receptor (EGFR) exon 20 insertion mutations. Read more
- FDA approved sotorasib (Lumakras™) as the first treatment for NSCLC patients whose tumors have the KRAS G12C genetic mutation and who have received at least one prior systemic therapy. Read more
March
- FDA approved lorlatinib (Lorbrena®) targeted therapy drug now for the first-line treatment of NSCLC patients with the ALK mutation. Read more
February
- FDA approved cemiplimab-rwlc (Libtayo®) immunotherapy drug for first-line treatment for NSCLC patients with high PD-L1 expression and who are not eligible for surgery or chemoradiation. Read more
- FDA approved trilaciclib (Cosela™), a drug that reduces chemotherapy-induced bone marrow suppression, for SCLC patients. Read more
- FDA approved tepotinib (Tepmetko®) for the treatment of metastatic non-small cell lung cancer (NSCLC) patients who have the MET exon 14 skipping alterations. Read more
2020
December
- FDA approved osimertinib (Tagrisso®) for adjuvant therapy for NSCLC patients who have undergone resection and have tumors positive for either EGFR exon 19 or exon 21 L858R. Read more
October
- FDA approved FoundationOne CDx companion diagnostic test to help identify NTRK+ solid tumor patients eligible for larotrectinib and to help identify ALK+ patients eligible for alectinib. Read more
- FDA approved “cobas EGFR mutation test v2” to identify NSCLC patients eligible for any of the EGFR inhibitor therapies, including those used to treat EGFR exon 19 and L858R deletions, as well as any EGFR therapies to come in the future. Read more
September
- FDA approved pralsetinib (Gavreto®) for people with metastatic non-small-cell lung cancer (NSCLC) who have the MET exon 14 mutation. Read more
August
- FDA approved Guardant360 CDx assay, a liquid biopsy (blood test) that also uses next-generation sequencing (NGS) technology to identify patients with the EGFR mutation. Read more
June
- FDA approved lurbinectedin (Zepzelca®) for the second-line treatment of patients with metastatic SCLC. Read more
May
- FDA approved combination ramucirumab and erlotinib for first-line treatment of NSCLC patients with the EGFR exon 19 or exon 21 mutations. Read More
- FDA approved combination nivolumab (Opdivo®) and ipilimumab (Yervoy®) for the first-line treatment of patients with PD-L1+ metastatic NSCLC. This combination can also be used as a first-line treatment in conjunction with chemotherapy for metastatic NSCLC. Read more
- FDA approved brigatinib (Alunbrig®) as a first-line treatment for metastatic non-small cell lung cancer (NSCLC) patients with the ALK mutation. Read More
- FDA approved atezolizumab (Tecentriq®) as a first-line treatment for metastatic non-small cell lung cancer (NSCLC) patients who have high PD-L1 expression. In conjunction with this approval, the FDA approved the diagnostic test Ventana PD-L1 assay. Read More
- FDA approved capmatinib (Tabrecta®) as a treatment for metastatic non-small cell lung cancer (NSCLC) patients who have the MET exon 14 mutation. In conjunction with this approval, the FDA approved the diagnostic test FoundationOne CDx assay. Read More
- FDA approved selpercatinib (Retevmo®) as a treatment for non-small cell lung cancer (NSCLC) patients or thyroid cancer patients who have the RET mutation. Read More
- FDA approved nivolumab (Opdivo®) plus ipilimumab (Yervoy®) and chemotherapy for first-line treatment of metastatic NSCLC
March
- FDA approved durvalumab (Imfinzi®) in combination with chemotherapy as a first line treatment for extensive stage small cell lung cancer (SCLC). Read More
February
- FDA approved pemetrexed (Pemfexy®) injectable chemotherapy for treatment of patients with advanced non-squamous non-small cell lung cancer (NSCLC). Read More
Approved drugs by decade
1940s
Mechlorethamine Hydrochloride
1970s
Doxorubicin Hydrochloride
Methotrexate Sodium
1980s
Cisplatin
1990s
Etoposide / Etoposide Phosphate
Gemcitabine Hydrochloride
Vinorelbine Tartrate
2000s
Bevacizumab
Carboplatin
Docetaxel
Erlotinib Hydrochloride
Everolimus
Gefitinib
nab-Paclitaxel
Pemetrexed Disodium
Topotecan Hydrochloride
2010s
Afatinib Dimaleate / Afatinib
Alectinib
Atezolizumab
Brigatinib
Ceritinib
Crizotinib
Dabrafenib Mesylate
Dacomitinib
Durvalumab
Entrectinib
Larotrectinib
Lorlatinib
Necitumumab
Nivolumab
Osimertinib Mesylate
Pembrolizumab
Ramucirumab
Trametinib Dimethyl Sulfoxide
2020s
Adagrasib
Amivantamab-vmjw
Binimetinib
Capmatinib Hydrochloride
Cemiplimab-rwlc
Encorafenib
Fam-trastuzumab deruxtecan-nxki
Ipilimumab
Lurbinectedin
Mobocertinib Succinate
Pralsetinib
Selpercatinib
Sotorasib
Tepotinib Hydrochloride
Tremelimumab
Trilaciclib